CURE-OP
Combinational Ultrasound Radiotherapy Enhanced Oncology Platform
The aim of CURE-OP is to develop the first commercially available High Intensity Focused Ultrasound (HIFU) platform specifically designed for combinational cancer therapy. We have developed a robotic device that can deliver a broad range of ultrasound regimes (hyperthermia, thermal ablation and cavitation), enabling different types of cancer polytherapy. During this project, the HIFU device is used in combination with a radiotherapy (RT) machine and tested clinically.
Surgeons and oncologists have been showing a keen interest in using HIFU to treat cancer, especially in combination with existing treatments. HIFU is a non-ionizing radiation, which can enhance RT treatments, thus increasing the safety profile and efficacy with reduced ionizing radiation and lower costs of the overall treatment. It is possible to explore other combinational therapies such as local drug delivery and immunotherapy with malignant tumour ablation.




Extracorporeal high intensity focused ultrasound (HIFU) provides a noninvasive alternative to conventional therapies. It consists of targeting a specific tissue area using a focused acoustic wave. While it goes through the tissue, the wave will lose some energy that will be dissipated in heat leading to a rise of temperature in the tissue.
Cancer has a significant and increasing impact on society as it was the second leading cause of death worldwide in 2018 with 9.6 million deaths estimated.
Radiotherapy (RT) is one of the most used cancer therapies. However, this technique induces serious side effects. Furthermore, the ionizing radiation delivery cannot be repeated several times due to lifetime dose accumulation. Heat is a known radiation sensitizer.
Indeed, hyperthermia (HT) is a therapeutic procedure often used as a cancer adjuvant therapy. It generally refers to the raise of the tissue temperature between 40 and 45 °C for several minutes up to an hour. Through different mechanisms such as immune stimulation or increase of perfusion and oxygenation, HT improves tumor responses to radiation therapy.
Combining RT and moderate heat could then lead to a reduction of ionizing radiation delivery improving patient safety and treatment efficiency. Thus, the idea to use HIFU to generate moderate heat in combination with RT. Theraclion is a French MedTech company specialized in echotherapy.
CURE OP‘s objective was to develop an ultrasound therapy prototype machine for combinational cancer therapy.
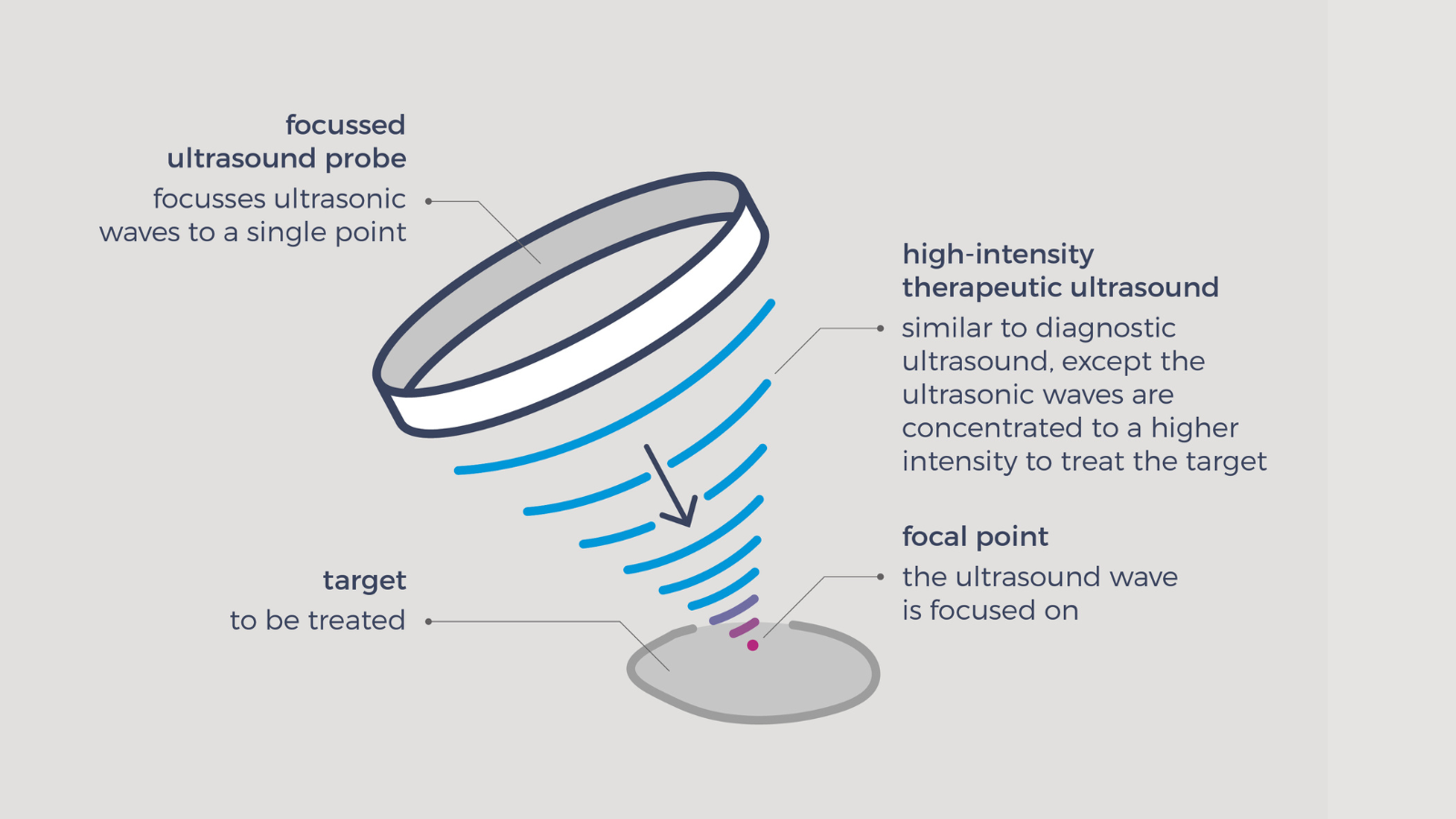
HIFU is focused ultrasound at intensities in the range of Isa = 100-10000W/cm2. Both mechanical and thermal effects can occur in most applications. The most common mechanical effect is cavitation and the classic thermal effect is heating, due to absorption. The geometry of the heated/ablated area is often described as cigar-shaped, matching the focal volume of the HIFU field. Depending on the transducer frequency-of-operation, the size can range from a few millimetres at higher frequencies, up to a couple of centimetres for low frequency treatments.
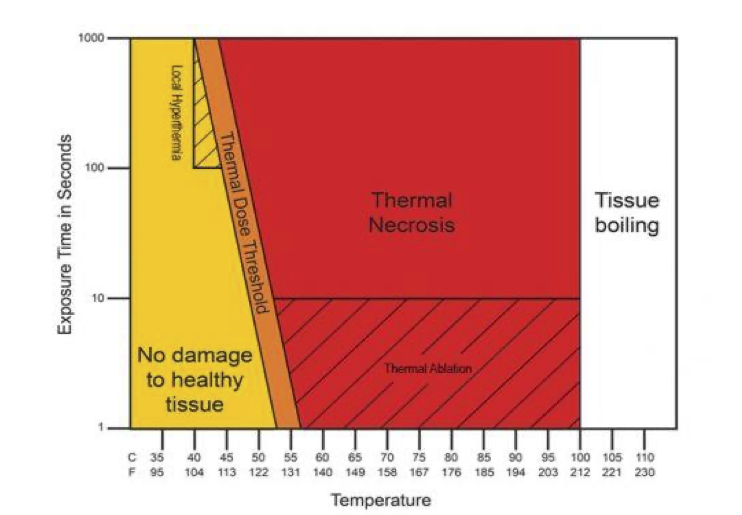
Hyperthermia (HT) is a therapeutic procedure that tends to raise the tissue temperature between 40 and 45 °C. This technique differs from thermal ablation as it doesn’t target direct necrosis through intense and rapid heat (> 60°C) but rather maintain a mild heat for a longer time. It is mostly used in combination with other therapies like chemotherapy (CT) and radiotherapy (RT). Its goal is to improve the clinical response and decrease oncological treatment toxicity by sensitizing tumors allowing the main treatment to treat radio resistant tumors and/or lower ionizing radiation delivered to patient. Moderate heat of tissue increases blood perfusion as well as tumor oxygenation. As hypoxia (oxygen deficit) is a major factor of radio resistance, those microenvironment changes could lead to better tumor responses.
Cancer has a significant and increasing impact on society as it was the second leading cause of death worldwide in 2018 with 9.6 million deaths estimated. Radiotherapy (RT) is one of the most used cancer therapies. However, this technique induces serious side effects. Furthermore, the ionizing radiation delivery cannot be repeated several times due to lifetime dose accumulation.

Theraclion is a French MedTech company specializing in high-tech therapeutic ultrasound equipment. We design and market Echopulse® and SONOVEIN®, two innovative echotherapy solutions.

ICCAS is part of the University of Leipzig in Germany. The Innovation Center Computer Assisted Surgery is a pioneer in developing computer-aided, integrative technologies and intelligent assistance systems in medicine. It is the interface between clinical requirements and economic implementation of innovative medical products. The main goal is the improvement of therapy methods and work processes to increase patient safety and economically relevant technology.

mediri is a German company specialized in clinical imaging, research and software development. Offering state-of-the-art technology for secure image upload and processing tailored to the individual customer needs, mediri has a strong research track tenure and is collaborating with Universities and research institutions in numerous scientific projects on advancing image analysis and diagnosis support.

Fraunhofer MEVIS is the German Institute for Digital Medicine. It develops real-world software solutions for image and data supported early detection, diagnosis, and therapy. Strong focus is placed on cancer as well as diseases of the circulatory system, brain, breast, liver, and lung. The goal is to detect diseases earlier and more reliably, tailor treatments to each individual, and make therapeutic success more measurable.
WORK PACKAGE 1
Project management and IPR management
The objective of this WP is to implement a project management framework to coordinate and monitor the project tasks, track budget vs. actual cost and follow-up achievements as well as to manage IPR issues.
WORK PACKAGE 2
Specifications
This WP aims at providing the required specifications related to the development of the hardware (structure of the device, components …) and software (structure design of the software, etc.). It will also be dedicated to the definition of the test and validation plan.
WORK PACKAGE 3
Hardware development
In this WP Theraclion will design and develop the mechanical and electronic hardware of the device according to the specifications defined in WP2:
1. The cabinet (including electronics, cooling system, power supplies, mechanical design, ultrasound engine): many components of the Echopulse system will be incorporated into the new device. Nevertheless, the firmware (embedded software to drive the hardware) and the user interface (UI) will be adapted to the modified hardware and 3D imaging software.
2. The interface to the Treatment Head (VTU)
3. Software interfaces (basic low-level software components for hardware control, therapy planning and treatment control)
4. The ultrasound engine (electronics for the ultrasound imaging subassembly) will be acquired from an OEM vendor.
WORK PACKAGE 7
HIFU pulse design and validation
This work package is entirely dedicated to investigate new pulse parameters that can be used to achieve mild heating or mild mechanical stress in tissue. Traditionally HIFU has been used for thermal ablation using highly energetic pulse sequences to achieve tissue coagulation. Theraclion uses a calibration technique which allows to fine tune the delivered acoustic power in order to assure reliable thermal ablation. We are planning to use a similar technique enhanced by numerical real-time simulations that can take advantage of a probing in order to optimize the energy delivery. Task 7.1 will define the parameters space and allow to validate to calibrate and validate the simulations with in controlled experiments. This experiments will be executed in ex-vivo tissue and in vivo animal models, which are already validated for ablation pulses. Task 7.2 will bring extra information to the simulation which can be run in real-time during a treatment. This information will be based on the attitude of tissue to change acoustical properties when the temperature changes. With fast processing times nowadays easily achievable the temperature of each adjacent pulse can be better estimated. The input is a probing or backscatter signal, which will be used to regulate the delivered power. Task 7.3 will be used to validate temperature control and generally the HIFU pulse and an in vivo setting to assure good translation into to the clinical trails.
WORK PACKAGE 8
Integration and clinical validation
The software and the hardware developed in WP 3 and 4 will be integrated together, then tested and validated in vivo.
WORK PACKAGE 9
Dissemination, regulary activities and market deployment
The objectives of WP6 are to have the new technologies resulting from the project interfaced with the different targeted data users (medical practitioner, industrialization….) and the corresponding market opportunities. To achieve this goal, the related tasks aim at: • Promoting extensively the project works in the medical sector, establishing links with related on-going research and industrial initiatives in hospitals and training centers (including university);
• Setting the foundations for future commercial exploitation and opportunities.
• Prepare the regulatory files to obtain the CE marking
WORK PACKAGE 6
Robotics and RT workflow
RT workflow integration, Synchronization, Robotic movement
The main purpose of this work package is the integration of the ultrasound therapy hardware with the robotic arm, the treatment head and the electronic cart into the radiation therapy facility. The challenge is here, that neither the ultrasound system should influence or hamper radiation therapy nor that the RT system influences the proper US therapy. Final goal is to allow a simultaneous US-RT-therapy. However, an intermediate stage would be a consecutive application of US followed by RT.
The work package is structured in 5 tasks. In Task 6.1 the physical hardware integration of the CURE-OR system is addressed. The CURE-OR system has to be fixed near the patient table so that both therapy modalities reach the target volume. In Task 6.2 a produce will be developed to synchronize the coordinate system of both therapy systems. Task 6.4 ensures that the robotic arm does not shield the beam of the RT system. Aim of Task 6.4 is to ensure a permanent acoustic coupling of the US treatment head to the skin of the patient, even if the treatment head moves due to focal steering of the HIFU beam or during the acquisition of diagnostic US images. The fine steering of the robotic arm in order to perform US-therapy (heating, ablation) or US imaging is solved in Task 6.5.
WORK PACKAGE 5
US Therapy planning and advanced imaging
This work package concentrates on planning of the ultrasound therapy as supplement to the RT-therapy planning. For this several data and information gained during the RT planning should be used for the US treatment. E.g. such data are CT and MR images, segmentation information of the different target volumes and the RT organs at risk. US therapy base on ultrasound image data from the CURE-OP treatment head (comp. T 6.5 and T4.2). So a fast and precise registration of this 3D data with the CT/ MR planning images is necessary. In addition, this WP also include experiments to assess whether photoacoustic imaging may yield added value for therapy monitoring in our setup.
The WP is structure in 6 tasks. In Task 5.1 the RT plan is analyzed and all information useful for US therapy planning will be automatically extracted. This is followed by the US therapy planning in task 5.2. Task 5.3 focus on the registration of the US data with the CT/MR data. The optimization task 5.4 allows to improvement the different software modules of WP 5.
WORK PACKAGE 4
Therapy visualisation and motion control
The objective of this WP is to provide software modules to operate the US-RT therapy with the CURO-OP hardware.
This work package is structured into five tasks. The goal of the first task is to collect all data, mainly ultrasound images and position data, and to provide these data for all other components of the software. In the second task the software for the reconstruction and registration of 3D image (3D volumes) from the 2D ultrasound images will be designed. A graphical user interface and visualization of imaging data will be done in the third task. An advance motion detection module with automated feature detection for the compensation of target motion will be developed in task 4.4. The objective of the last task 4.5 is to optimize the software after first completion and integration of all software components. In this task bugs and shortcomings of the first implementation will be fixed and the software improved.









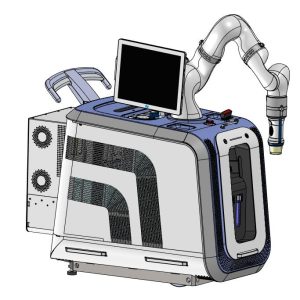
The objective of this project was to develop the first commercially available high intensity focused ultrasound (HIFU) platform specifically designed for combinational cancer therapy. See photo below. We developed a robotic device that can deliver a broad range of ultrasound regimes, enabling different types of cancer polytherapy. In the scope of this project’s timeframe, the HIFU device was designed in order to be used in combination with a radiotherapy (RT) machine. Surgeons and oncologists have been showing a keen interest in using HIFU to treat cancer, especially in combination with existing treatments. HIFU is a non-ionizing radiation, which can enhance RT treatments, thus increasing the safety profile and efficacy with reduced ionizing radiation and lower costs of the overall treatment. It will be possible to explore other combinational therapies such as local drug delivery and immunotherapy with malignant tumour ablation.
With CURE-OP we developed an ultrasound therapy system designed for combinational cancer therapy, with an initial validation on head and neck cancer or breast cancer or melanoma. Further perspectives to apply combinational treatment to other solid cancers will be assessed.
Radiation therapy (RT) is one of the most used cancer therapies. It is currently used as one of the major curative treatments for breast cancer. Unfortunately, there can be serious short and long-term side effects, and above all, treatments cannot be repeated several times.
High intensity focused ultrasound (HIFU) is very precise and non-invasive: no general anaesthesia, no scars. It has been used for several years in the treatment of certain tumours (breast fibroadenoma, prostate cancer, uterine fibroids, thyroid nodules). The ultrasonic energy quickly heats the tissue locally. Heat is known to sensitize tissue to radiation.
Theraclion is currently involved in a feasibility (proof of concept) clinical study at University of Virginia hospital to enhance immunotherapy, these studies use a device which includes some of the subsystems developed during this project. This alone has been a tremendous outcome of this project. We proceeded in the development of the CURE-OP prototype developing several new subsystems, which are described below. The advantage of developing each subsystem separately, made it possible to introduce some of them as early as possible into other Theraclion products and leveraging even more the outcome of this project.
The device prior to this project was not adapted for cancer therapy and we addressed in this project many of the limitations such as: complete lesion coverage is very difficult to achieve; absence of image registration to other image modalities; sub-ablational regimes are not available and cannot be monitored.
With CURE-OP we wanted to create a device which is flexible and that can deliver and monitor optimized treatments (sub-ablative regimes) based on the best available imaging information and improved target access.
In a nut shell and without going too much in technical details we completely redesigned: the high intensity focussed ultrasound engine – developing a new HIFU transducer and a versatile radio frequency (RF) generator; we integrated one of the best state of the art diagnostic ultrasound scanners; we replaced the positioning arm with the best medical cobot available on the market; we integrated a dedicated (MIC – Many Integrated Cores) computer to run the newly developed thermometry algorithm. Furthermore we added a new degassing system and completely changed the cooling system for the EPack™, ensuring patient cooling and coupling functions.
Finally to make all these subsystems work in syntony we updated and improved all levels of Theraclions user software (TUS) and its components.
The result of several years of work is the final CURE-OP prototype which is currently being certified for human use. Deliverable as described in the updated proposal have been achieved; for more details on all deliverables please see the report attached to this form.
| Milestone | type | description | status |
| D1.1 | document | PPR | yes |
| D1.2 | document | Exploitation plan | yes |
| D2.1 | document | hardware specifications | yes |
| D2.2 | document | software specifications | yes |
| D2.3 | document | validation plan | yes |
| D2.4 | document | subcontractor Robot | yes |
| D2.5 | document | RT manuf | yes |
| D3.1 | document | system design | yes |
| D3.2 | document | mechanical interface design | yes |
| D3.3 | document | electronic interface design | yes |
| D3.4 | document | robot software interface spec | yes |
| H3.1 | hardware | HIFU cabinet functional | yes |
| H3.2 | hardware | mechanical interface parts | yes |
| H3.3 | hardware | electronic interface | yes |
| S3.4 | software | HIFU to robot software interface and UI | yes |
| S7.1 | software | Improved algorithms and temperature predictions | yes |
| D7.2 | document | method to estimate the temperature | yes |
| D7.3 | document | method to deliver the correct pulse | yes |
| D8.2 | document | Platform tested functionally | yes |
| D8.3 | document | Platform tested | yes |
| D9.1 | document | CURE-OP Website | yes |
| D9.2 | document | Report on dissemination activities | yes |
| D9.3 | document | Project business plan including steps for market deployment | yes |
Transducer design
We developed a high efficient dual frequency HIFU transducer (1 and 3 MHz), for deep and superficial targets.
Generator
The developed a compact and easily to manufacture RF generator specifically designed to for very high power multi frequency operations.
Degassing
We developed a miniaturized degassing system to cope with high intensity bursts required in this project.
Robot
We integrated a state of the art 7-axis medical grade robot to enable maximal flexibility. This will give the necessary additional flexibility in positioning for an optimal acoustic access to the target volume. This will also speed up therapy procedure. Non-invasive, precise, fast and confluent tumour exposure requires highly sophisticated image guidance and monitoring. For the first time, combinational therapy of RT and HIFU will be clinically available for the treatment of superficial cancers, such as head and neck breast and melanoma.
Software for new device
We adapted the existing control software to accommodate seeimeslyseamlessly the new robot and other components.
Treatment pulse
We designed a pulse for hyperthermia for a combinational treatment with radiotherapy. A pulse following a linear trajectory (five consecutive lines) with continuous 30-second shooting at 1 MHz was evaluated. Numerical simulations in a homogeneous medium (soft tissue) demonstrated that this pulse could generate temperature between 50 and 54 °C without exceeding the temperature threshold (55 °C) and deliver enough thermal dose (above 1200 seconds at 43 °C). An ex vivo experiment was designed using four thermocouples to measure temperature evolution in turkey breast. Maximal temperatures measured were between 55 and 60 °C which represents about 10 % difference with spike amplitude in numerical simulations. The heterogeneity of real tissue as well as thermocouples artefacts probably lead to the quantitative differences between simulations and ex vivo experiments.
Thermometry
HIFU can heat the tissues but monitoring the temperature on ultrasound images remains a challenge. A “speckle tracking” algorithm is used to estimate the temperature using an “echo shift” model. Indeed, in the field of hyperthermia, the temperature is proportional to the derivative of the tissue displacement. Thanks to pre and post pulse images, the algorithm provides the temperature. After ex-vivo calibration, the results of the algorithm are compared with values measured by thin thermocouples. The difference between the thermocouples was tested for 4 different powers and is on average ±0.5°C.
